You Develop the Tlc Plate Again and Are Able to Visualize 3 Spots
Thin Layer Chromatography (TLC)
TLC is a simple, quick, and inexpensive procedure that gives the chemist a quick answer as to how many components are in a mixture. TLC is besides used to support the identity of a compound in a mixture when the Rf of a compound is compared with the Rf of a known compound (preferably both run on the same TLC plate).
A TLC plate is a canvass of glass, metal, or plastic which is coated with a thin layer of a solid adsorbent (usually silica or alumina). A small corporeality of the mixture to be analyzed is spotted near the bottom of this plate. The TLC plate is so placed in a shallow pool of a solvent in a developing bedchamber and so that simply the very bottom of the plate is in the liquid. This liquid, or the eluent, is the mobile phase, and it slowly rises up the TLC plate by capillary action.
As the solvent moves past the spot that was applied, an equilibrium is established for each component of the mixture betwixt the molecules of that component which are adsorbed on the solid and the molecules which are in solution. In principle, the components will differ in solubility and in the strength of their adsorption to the adsorbent and some components will be carried further up the plate than others. When the solvent has reached the meridian of the plate, the plate is removed from the developing sleeping accommodation, stale, and the separated components of the mixture are visualized. If the compounds are colored, visualization is straightforward. Commonly the compounds are not colored, and then a UV lamp is used to visualize the plates. (The plate itself contains a fluorescent dye which glows everywhere except where an organic compound is on the plate.)
How To Run a TLC Plate
 | Pace 1: Set up the developing containerThe developing container for TLC tin can exist a specially designed chamber, a jar with a chapeau, or a beaker with a lookout man glass on the top (the latter is used in the undergrad labs at CU). Cascade solvent into the bedchamber to a depth of just less than 0.5 cm. To assistance in the saturation of the TLC bedchamber with solvent vapors, you can line part of the within of the beaker with filter paper. Cover the beaker with a watch glass, swirl it gently, and allow it to stand while y'all prepare your TLC plate. |
 | Step two: Prepare the TLC plateTLC plates used in the organic chem teaching labs are purchased as five cm ten 20 cm sheets. Each large canvass is cut horizontally into plates which are 5 cm tall by diverse widths; the more samples y'all plan to run on a plate, the wider it needs to be. Shown in the photo to the left is a box of TLC plates, a big un-cut TLC sail, and a small TLC plate which has been cut to a user-friendly size. Handle the plates carefully so that you do not disturb the blanket of adsorbent or become them muddy. |
 | Measure out 0.5 cm from the bottom of the plate. Using a pencil, depict a line across the plate at the 0.five cm mark. This is the origin: the line on which you volition spot the plate. Take intendance not to press and so difficult with the pencil that y'all disturb the adsorbent. Under the line, marking lightly the proper noun of the samples y'all volition spot on the plate, or mark numbers for fourth dimension points. Leave enough infinite between the samples then that they do non run together; almost 4 samples on a 5 cm wide plate is advised. |
 | Step 3: Spot the TLC plateIf the sample is not already in solution, deliquesce about ane mg in 1 mL of a volatile solvent such as hexanes, ethyl acetate, or methylene chloride. Every bit a rule of thumb, a concentration of i% usually works well for TLC analysis. If the sample is besides concentrated, it will run as a smear or streak (see troubleshooting section below); if it is not concentrated plenty, yous will see nothing on the plate. Sometimes you will need to use trial and mistake to get well-sized, easy to read spots. |
 | Obtain a a microcapillary. In the organic pedagogy labs, nosotros use 10µL microcaps - they are easier to handle than the smaller ones used in research labs. Dip the microcap into the solution and then gently affect the end of it onto the proper location on the TLC plate. Don't allow the spot to become likewise big - if necessary, y'all can touch information technology to the plate, lift it off and accident on the spot. If you repeat these steps, the wet expanse on the plate will stay small. |
 | This case plate has been spotted with iii unlike quantities of the same solution and is prepare to develop. If you lot are unsure of how much sample to spot, you tin can always spot multiple quantities and see which looks best. |
 | Step iv: Develop the plateIdentify the prepared TLC plate in the developing beaker, cover the beaker with the watch glass, and leave it undisturbed on your bench top. The solvent will rise up the TLC plate by capillary activity. Make certain the solvent does non cover the spot. |
 | Let the plate to develop until the solvent is about half a centimeter below the tiptop of the plate. Remove the plate from the beaker and immediately mark the solvent front with a pencil. Allow the plate to dry out. |
 | Stride five: Visualize the spotsIf in that location are whatsoever colored spots, circle them lightly with a pencil. Well-nigh samples are not colored and need to be visualized with a UV lamp. Concur a UV lamp over the plate and circumvolve any spots you lot see. Beware! UV calorie-free is dissentious both to your eyes and to your skin! Make sure you are wearing your goggles and practice not look directly into the lamp. Protect your skin by wearing gloves. |
 | If the TLC plate runs samples which are too concentrated, the spots will exist streaked and/or run together. If this happens, you volition have to beginning over with a more dilute sample to spot and run on a TLC plate. |
 | Here's what overloaded plates look similar compared to well-spotted plates. The plate on the left has a large yellowish smear; this smear contains the same 2 compounds which are nicely resolved on the plate side by side to it. |
TLC Solvents Choice
When yous need to determine the best solvent or mixture of solvents (a "solvent system") to develop a TLC plate or chromatography cavalcade loaded with an unknown mixture, vary the polarity of the solvent in several trial runs: a process of trial and error. Carefully detect and record the results of the chromatography in each solvent system. You lot volition find that as you increase the polarity of the solvent organization, all the components of the mixture move faster (and vice versa with lowering the polarity). The platonic solvent organisation is merely the organization that gives the best separation.
TLC elution patterns usually carry over to cavalcade chromatography elution patterns. Since TLC is a much faster process than cavalcade chromatography, TLC is ofttimes used to decide the best solvent system for cavalcade chromatography. For instance, in determining the solvent system for a flash chromatography procedure, the ideal system is the one that moves the desired component of the mixture to a TLC Rf of 0.25-0.35 and will separate this component from its nearest neighbor by difference in TLC Rf values of at to the lowest degree 0.20. Therefore a mixture is analyzed by TLC to determine the ideal solvent(s) for a flash chromatography procedure.
Beginners often do non know where to start: What solvents should they pull off the shelf to use to elute a TLC plate? Because of toxicity, price, and flammability concerns, the common solvents are hexanes (or petroleum ethers/ligroin) and ethyl acetate (an ester). Diethyl ether can be used, but it is very flammable and volatile. Alcohols (methanol, ethanol) can be used. Acetic acid (a carboxylic acrid) can be used, usually equally a small per centum component of the system, since it is corrosive, not-volatile, very polar, and has irritating vapors. Acetone (a ketone) can be used. Methylene chloride or and chloroform (halogenated hydrocarbons) are proficient solvents, only are toxic and should exist avoided whenever possible. If two solvents are equal in operation and toxicity, the more volatile solvent is preferred in chromatography considering it will be easier to remove from the desired compound after isolation from a column chromatography procedure.
Ask the lab teacher what solvents are available and advisable. And so, mix a non-polar solvent (hexanes, a mixture of 6-carbon alkanes) with a polar solvent (ethyl acetate or acetone) in varying percent combinations to make solvent systems of greater and lesser polarity. The charts below should aid you in your solvent option. You lot tin can too download this pdf chart of elution club. 

Interactions Between the Compound and the Adsorbent
The force with which an organic compound binds to an adsorbent depends on the forcefulness of the following types of interactions: ion-dipole, dipole-dipole, hydrogen bonding, dipole induced dipole, and van der Waals forces. With silica gel, the dominant interactive forces between the adsorbent and the materials to be separated are of the dipole-dipole type. Highly polar molecules interact fairly strongly with the polar SiOH groups at the surface of these adsorbents, and will tend to stick or adsorb onto the fine particles of the adsorbent while weakly polar molecules are held less tightly. Weakly polar molecules more often than not tend to move through the adsorbent more than quickly than the polar species. Roughly, the compounds follow the elution society given above.
The Rf value
The retention factor, or Rf, is defined equally the altitude traveled by the chemical compound divided by the altitude traveled by the solvent.

For case, if a chemical compound travels two.1 cm and the solvent front travels ii.8 cm, the Rf is 0.75:
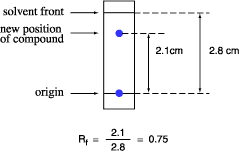
The Rf for a compound is a constant from one experiment to the next merely if the chromatography conditions below are likewise abiding:
- solvent organization
- adsorbent
- thickness of the adsorbent
- amount of material spotted
- temperature
Since these factors are difficult to keep constant from experiment to experiment, relative Rf values are by and large considered. "Relative Rf" means that the values are reported relative to a standard, or it ways that you compare the Rf values of compounds run on the same plate at the aforementioned time.
The larger an Rf of a compound, the larger the distance it travels on the TLC plate. When comparing two unlike compounds run under identical chromatography atmospheric condition, the compound with the larger Rf is less polar because information technology interacts less strongly with the polar adsorbent on the TLC plate. Conversely, if you know the structures of the compounds in a mixture, you can predict that a compound of low polarity will have a larger Rf value than a polar compound run on the same plate.
The Rf can provide corroborative evidence as to the identity of a compound. If the identity of a compound is suspected simply not nevertheless proven, an authentic sample of the chemical compound, or standard, is spotted and run on a TLC plate side by side (or on top of each other) with the compound in question. If two substances have the same Rf value, they are likely (but not necessarily) the same compound. If they have unlike Rf values, they are definitely different compounds. Notation that this identity check must be performed on a unmarried plate, because it is difficult to duplicate all the factors which influence Rf exactly from experiment to experiment.
Troubleshooting TLC
All of the to a higher place (including the procedure folio) might sound like TLC is quite an easy process. Only what well-nigh the first time you lot run a TLC, and see spots everywhere and blurred, streaked spots? As with whatsoever technique, with practice you get amend. Examples of mutual problems encountered in TLC:
- The compound runs every bit a streak rather than a spot: The sample was overloaded. Run the TLC again later diluting your sample. Or, your sample might but comprise many components, creating many spots which run together and appear as a streak. Perhaps, the experiment did not become as well every bit expected.
- The sample runs as a smear or a upward crescent: Compounds which possess strongly acidic or basic groups (amines or carboxylic acids) sometimes show up on a TLC plate with this behavior. Add a few drops of ammonium hydroxide (amines) or acetic acid (carboxylic acids) to the eluting solvent to obtain clearer plates.
- The sample runs as a downwards crescent: Probable, the adsorbent was disturbed during the spotting, causing the crescent shape.
- The plate solvent front end runs crookedly: Either the adsorbent has flaked off the sides of the plate or the sides of the plate are touching the sides of the container (or the paper used to saturate the container) as the plate develops. Kleptomaniacal plates go far harder to measure Rf values accurately.
- Many random spots are seen on the plate: Make sure that you do not accidentally drop any organic compound on the plate. If get a TLC plate and get out it laying on your workbench as you do the experiment, you might drop or splash an organic compound on the plate.
- You see a mistiness of bluish spots on the plate equally it develops: Perhaps you used an ink pen instead of a pencil to marking the origin?
- No spots are seen on the plate: You might not have spotted plenty chemical compound, mayhap because the solution of the compound is besides dilute. Try concentrating the solution, or spot it several times in i identify, allowing the solvent to dry between applications. Some compounds do not show upward under UV light; try another method of visualizing the plate (such every bit staining or exposing to iodine vapor). Or, perhaps you do not take any compound considering your experiment did not go every bit well as planned. If the solvent level in the developing jar is deeper than the origin (spotting line) of the TLC plate, the solvent will dissolve the compounds into the solvent reservoir instead of allowing them to motility upward the plate past capillary action. Thus, you will not see spots later the plate is developed. These photos show how the yellow compound is running into the solvent when lifted from the developing jar.

TLC Technique Quiz
See how well you understand TLC by taking the online TLC Technique Quiz!
Source: https://www.orgchemboulder.com/Technique/Procedures/TLC/TLC.shtml
0 Response to "You Develop the Tlc Plate Again and Are Able to Visualize 3 Spots"
Post a Comment